Chronicity of Viral Infections (CVI)

Presentation
 The Chronicity of Viral Infections [CVI] is an Emerging Team investigating the molecular and cellular mechanisms driving pathogen persistence in myeloid cells. Our goals are: i) to think about strategies to neutralize pathogens before they reach safe intracellular niches (such as the macrophage and other myeloid cells, including their hematopoietic progenitors) and also to think about how to block the pathogen spreading from these cellular niches to different cells and tissues; ii) to unveil the mechanisms of latency of intracellular pathogens, responsible for disease persistence; and iii) to contribute to the development of efficient therapeutic approaches for chronic, potentially lethal, diseases. Currently we are tackling long-term immune dysfunctions caused by HIV-1 and SARS-CoV-2 hosted by macrophages, megakaryocytes and their precursors, in order to understand these myeloid viral hideouts and ultimately devise improved therapeutic strategies against HIV/AIDS and COVID-19.
The Chronicity of Viral Infections [CVI] is an Emerging Team investigating the molecular and cellular mechanisms driving pathogen persistence in myeloid cells. Our goals are: i) to think about strategies to neutralize pathogens before they reach safe intracellular niches (such as the macrophage and other myeloid cells, including their hematopoietic progenitors) and also to think about how to block the pathogen spreading from these cellular niches to different cells and tissues; ii) to unveil the mechanisms of latency of intracellular pathogens, responsible for disease persistence; and iii) to contribute to the development of efficient therapeutic approaches for chronic, potentially lethal, diseases. Currently we are tackling long-term immune dysfunctions caused by HIV-1 and SARS-CoV-2 hosted by macrophages, megakaryocytes and their precursors, in order to understand these myeloid viral hideouts and ultimately devise improved therapeutic strategies against HIV/AIDS and COVID-19.
Research Activity
The research program of the Chronicity of Viral Infections [CVI] Emerging Team is currently divided in two research axes :
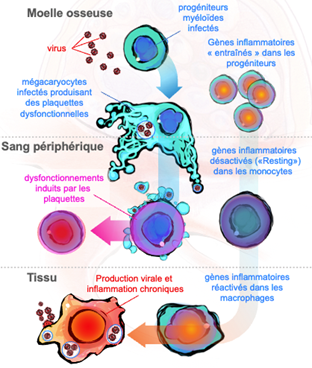
- Macrophages are durable hosts for viral pathogens via infection of common myeloid progenitors
Chronic inflammation is commonly observed in HIV-infected patients despite antiretroviral treatment and in individuals with post-acute sequelae of COVID-19. We are investigating: i) whether macrophages precursors (common myeloid progenitors) are primed early at the bone marrow stage by a first inflammatory stimulus (e.g. viral primo-infection), displaying thereafter an enhanced inflammatory response upon a second stimulus (e.g. re-infection or superinfection) once they are terminally differentiated into tissue-like macrophages; and ii) whether this trained response is directly implicated in persistent viral replication in tissular macrophages, sustaining infection and chronic inflammation deleterious for the homeostasis.
- Megakaryocytes targeted by viruses participate in infectious disease progression
Megakaryocytes, precursor of platelets, are gaining momentum in immunology field as inflammatory/immune system actors – a poorly understood but exciting topic in immunology. We will investigate the detrimental consequences of megakaryocyte infection for the immune system and the cellular/molecular mechanisms controlling viral persistent in this myeloid cell. We aim at deciphering the megakaryocyte factors expressed upon viral infections that will be inherited by their daughter platelets and that thereafter will immunomodulate immune cells upon platelet-cell contact. Virus-induced, platelet druggable targets will be functionally assessed by blockage/inhibition using antibodies and CRISPR/Cas9 strategies, in in vitro and in vivo models, opening avenues for novel therapeutical strategies.
Recent discoveries
Despite undetectable plasma viral load as a result of current antiretroviral therapy, HIV-infected individuals with poor immune reconstitution harbor infectious HIV within platelets. The presence of HIV in platelets is strongly correlated with poor immune reconstitution despite therapy. Megakaryocytes, in which proviral DNA and viral RNA were detected in these individuals, appear to be the likely origin of HIV-containing platelets. To investigate the mechanisms by which megakaryocytes support HIV-1 infection, we established in vitro models of viral infection in megakaryocytes derived from human CD34+ hematopoietic stem cells and in megakaryocyte cell lines. The integration of HIV proviral DNA into the megakaryocyte cell genome, self-limited viral production and HIV accumulation in virus-containing compartments – all features illustrating viral infection and HIV persistence within myeloid cells – have been established.
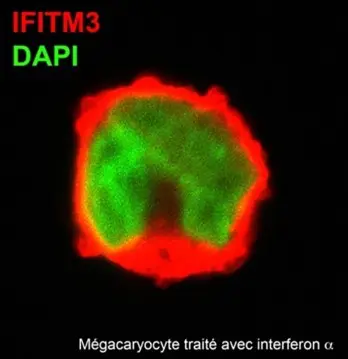
The team is investigating the mechanisms of virus-induced defusal of megakaryocytes’ antiviral factors, in particular the hijacking of the immune response mediated by interferon-induced transmembrane protein 3 (IFITM3). IFITM3 is an antiviral factor constitutively expressed by megakaryocytes, whose expression is enhanced by interferon alpha. IFITM3 expression was inhibited by HIV infection, without affecting the maturation of infected megakaryocytes. To investigate the involvement of IFITM3 in HIV infection, IFTIM3 gene expression was knocked-down using gene silencing techniques. As effect of IFTIM3 silencing, HIV infection is enhanced in megakaryocytes. Overall, these results suggest that HIV counteract the intrinsic antiviral response conferred on megakaryocytes by IFITM3. Megakaryocytes and platelets are largely neglected hideouts of HIV that deserve to be studied for the development of improved antiretroviral therapies and, above all, to achieve complete eradication of HIV infection.
Team Members
Fernando REAL
CNRS CRCN, responsable de l’équipe
Numéro ORCID : 0000-0002-5060-4334
Dylan JUCKEL
Post-doctorant (Université de Lille)
Agathe BOUR
Étudiante en Master 2 (Université de Lille)
Anaïs DARDAILLON
Doctorante (Université de Lille / CIFRE)
Cyrine BENTALEB
Post-doc (Université de Lille)
Noëmie COURTIN
Assistante Ingénieure (CNRS)
Selected publications
Bentaleb C, Adrouche S, (…) Real F*.
HIV-1 inhibits IFITM3 expression to promote the infection of megakaryocytes
Journal of Molecular Cell Biology. 2024 Oct 1, doi: 10.1093/jmcb/mjae042
Real F, Zhu A, Belmellat A, Sennepin A, (…), Bomsel M.
S100A8-mediated metabolic adaptation controls HIV-1 persistence in macrophages in vivo
Nature Communications. 2022 doi: 10.1038/s41467-022-33401-x
Real F, Ganor Y, Bomsel M.
Experimental Models to Study HIV Latency Reversal from Male Genital Myeloid Cells.
Methods in Molecular Biology. HIV reservoirs . 2022 doi: 10.1007/978-1-0716-1871-4_14
Real F, Capron C, Sennepin A, Arrigucci R, Zhu A, (…) Bomsel M.
Platelets from cART-suppressed HIV-infected patients with poor CD4+ T cell recovery carry infectious HIV.
Science Translational Medicine. 2020 doi: 10.1126/scitranslmed.aat6263
Pessoa CC, Reis LC, (…) Real F*.
ATP6V0d2 controls Leishmania parasitophorous vacuole biogenesis via cholesterol homeostasis
PLoS Pathogens. 2019 Jun 14;15(6):e1007834. doi: 10.1371/journal.ppat.1007834
Real F, Sennepin A, Ganor Y, Schmitt A, Bomsel M.
Live Imaging of HIV-1 Transfer across T Cell Virological Synapse to Epithelial Cells that Promotes Stromal Macrophage Infection.
Cell Reports. 2018 May 8;23(6):1794-1805. doi: 10.1016/j.celrep.2018.04.028
Keywords
Viral infections, Virus, Innate immunity, Macrophages, Megakaryocytes, Platelets, Immune dysfunction, Immunological failure, HIV/AIDS, SARS-CoV-2/COVID-19
Contact
Team contact (preferred)
03 20 87 77 98
Or
Fernando Real
Head of the Emerging team « Chronicity of Viral Infections »
fernando.real@cnrs.fr
03 20 87 78 02

