Université de Lille – CNRS UMR 9017 – Inserm U1019 – Institut Pasteur de Lille – CHU de Lille
With a size smaller than 10 kDa and a stable tertiary structure, miniproteins are complementary in many aspects to small molecules or large protein scaffolds for drug development. An asset of miniproteins is that they can be accessed through chemical synthesis, which enable introducing a large diversity of modifications, including those that are not managed by living systems. The increasing interest of the pharma industry for miniproteins is also stimulated by the significant advances in the chemical synthesis of proteins, an innovation process to which our team has been contributing significantly since its creation.
The miniproteins initially studied by the team were designed starting from human proteins. This part of the CBF project continues with the development of K1K1 Met tyrosine kinase agonist, and promising outcomes in the field of regenerative medicine (CNRS and SATT press releases). Very recently, the team expanded its approach by mining proteins produced by some worms that represent a rich source of miniproteins endowed with potent biological activities. The worms we are studying have evolved for more than 400 million years in diverse habitats or hosts. These extreme adaptations promoted the production of original active molecules that we are discovering and exploring for medical applications.
In parallel, we are strongly involved in the conception and validation of innovative protein production methods and modification tools. This work isstrongly connected to the others since it multiplies the possibilities to study the mechanisms of action of our miniproteins through chemical biology approaches, as well as to improve them according to the targeted specifications.
Projets
Nature-inspired innovation in chemical protein synthesis (O. Melnyk, V. Agouridas, V. Diemer)
Modern chemical protein synthesis primarily relies on the selective formation of peptide bonds by reacting unprotected peptide segments in aqueous media. Our aim is to develop innovative, bio-inspired and generally applicable chemical concepts to significantly broaden the scope of amide chemistry for protein assembly. In doing so, our findings will be of interest and practical value to a wide audience.
We have recently begun to apply this vision to the native chemical ligation (NCL). NCL is by far the most widely used peptide ligation method for protein semi or total synthesis up to now. This reaction consists of combining a peptide thioester with a cysteinyl (Cys) peptide to produce a larger peptide with a peptide bond to cysteine. However, and after more than 30 years of improvements, the scope of NCL for protein semi or total synthesis is still limited by:
- the low frequency of cysteine residues in proteins, which limits the types of junctions that can be made,
- the modest rate of peptide bond formation making protein assembly a lengthy process,
- the variable solubility of the peptide segments that are linked together,
- the paucity of biological methods for accessing peptide thioester reactants, a problem exacerbated by their restriction to C-terminal thioesters only
- the failure of NCL to be of value for peptide oligomerisation, and thus for the synthesis of peptide-based biomaterials
Very recent discoveries by the team provide a strong basis for addressing these issues (see Figure below, and more by cicking on the image). For example, we discovered that the introduction of charged amino acids (e.g. phosphoserine, arginine) into the peptide reactants can force their encounter and thus considerably boost the rate of chemical reactions. Building on these pioneering studies and others, our goal is to invent novel chemical methods enabling to overcome the above mentionned limitations.
This project is another demonstrative example of the power of chemical synthesis to unravel complex biological mechanisms, and of the interest of invertebrate models as a bio-inspiring source of original protein scaffolds.
It focuses on Venus Kinase Receptors (VKRs), an atypical class of receptor tyrosine kinases (RTK) discovered by J. Vicogne during his PhD on S. mansoni Tyrosine Kinases (TK). VKRs have a TK domain similar to that of the Insulin receptor and an extracellular domain containing a Venus Fly Trap (VFT) module. An arginine-induced rearrangement of the VFT conformation leads to the autophosphorylation of the TK domains and to the activation of a specific signaling pathway. VKRs are also present in many other invertebrate speciesincluding the mollusk Biomphalaria glabrata, the intermediate host of S. mansoni. In contrast, VKR are absent from its definitive human host as well as in any other vertebrate threads. VKRs play an important role in the development and reproduction of many invertebrates, including S. mansoni.
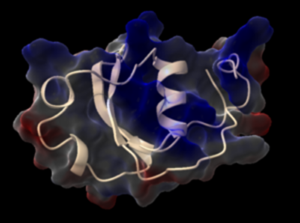
We identified a miniprotein produced by the mollusk host that interact with the VKRs of both the mollusc and the parasite S. mansoni. We could produce this protein by chemical synthesis and determine its X-ray crystal structure (see Figure below). This protein has opposite effect on host and parasite VKRs and might play a crucial role in host-parasite relationships. This miniprotein and its VKR receptors are currently the subject of intense research to decipher their binding interface through the determination of the structure of hormone-receptor complexes, and to understand their biological significance.
This WP focuses on the discovery and potentiation of cysteine-rich antimicrobial peptides (AMPs) from unique deep-sea worms as promising antibiotics against multidrug resistant (MDR) microorganisms such as Pseudomonas aeruginosa and Acinetobacter baumannii, which represent a major global public health threat. The emergence of MDR strains, mainly associated with pulmonary infections, together with ineffective and expensive therapeutics, urgently requires the development of a new generation of antimicrobials that can act through different mechanisms of action, thus offering new therapeutic options. The multi-target interaction of AMPs with the bacterial membrane makes the occurrence of AMR/MDR to AMPs more difficult compared to conventional antibiotics. Additional bioactivities, such as antibiofilm and anti-inflammatory activities, also add value to AMPs compared to conventional antibiotics. By comparing available sequences, the team was astonished to discover that unused compounds from worms living in deep-sea hydrothermal vents have natural mutations associated with physicochemical and biological properties that have taken terrestrial compounds dozens of years of laboratory optimisation before they could be used in industry.
The animals that inhabit deep-sea hydrothermal vents have an extraordinary physiology with under-explored and unique bioactive molecules that have been naturally designed and selected through millions of years of evolution to function regardless of extreme and highly variable conditions (salt, anoxia, pH 3 to 8, temperature from 4 to > 100 °C) to face high densities of Gram-negative bacteria, including bacteria of the genus Pseudomonas. Of the thousands of AMPs identified to date (mostly from soil bacteria or terrestrial animals such as insects, mammals and amphibians), only a few have reached the market due to production limitations, cytotoxic issues and reduced activity in clinically relevant environments (e.g. pH, salts, proteases). This clearly demonstrates that resistance to physiological conditions is a bottleneck in the development of AMPs and needs to be addressed in the development of a new therapeutic peptide.
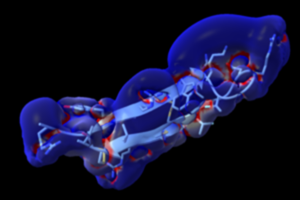
The high likelihood of discovering new bioactive extremophile AMPs that are effective in vivo, is already illustrated by alvinellacin (see Figure below), a patented AMP identified by A. Tasiemski from the emblematic worm Alvinella pompejana, for which promising data have been obtained with the CBF team and with Muriel Pichavant (Openfield Team). The AMP, which can be produced by chemical synthesis, has all the hallmarks of a promising drug for the treatment of P. aeruginosa infections. It is currently the subject of extensive optimizations and structural, biophysical and biological investigations.
Membres
CABY Stéphanie
Engineer, Institut Pasteur de Lille (IE)
DESMET Rémi
Technician, Institut Pasteur de Lille
DI ADAMO Julie
PhD student
DJEMAT-PARMENTIER Aurélie
CNRS Animal technician
HOUSSIER Robin
Student
LANCELOT Julien
Post-doc
MOUGEL Alexandra
CNRS Engineer (IEHC)
OLLIVIER Nathalie
Engineer Institut Pasteur de Lille, IR2
ROY Eliott
PhD student
SENECHAL Magalie
CNRS Assistant Engineer
SNELLA Benoît
Technician, Institut Pasteur de Lille
TERZANI Francesco
PhD student
WANG Chen
PhD student



